WASHINGTON, APRIL 19, 2026 —
Key Takeaways
- A landmark study of 175,000 women — the largest NHS breast cancer screening trial ever conducted — found that Google’s AI mammography system detected more cancers, fewer false positives, and 25% of cancers that human radiologists missed between scheduled screenings.
- The AI system reduced radiologist workload by nearly one-third, and performed particularly well for women having their first ever mammogram — detecting 8.8% more cancers while generating 39% fewer unnecessary recalls that cause anxiety and additional testing.
- MIT researchers simultaneously unveiled an AI system called CleaveNet that designs cancer-detecting molecular sensors with near-perfect specificity — an advance aimed at an at-home test that could eventually screen for 30 different cancer types from a urine sample.
Two separate artificial intelligence research breakthroughs published this week have advanced what scientists increasingly describe as the most consequential medical technology revolution of the generation: the use of AI to detect cancer earlier, more accurately, and more equitably than any previous screening method.
Taken together, the studies represent a shift from AI as a promising experimental tool to AI as a validated clinical asset — one that, in the right setting, is now demonstrably better than the trained human physicians it was designed to assist.
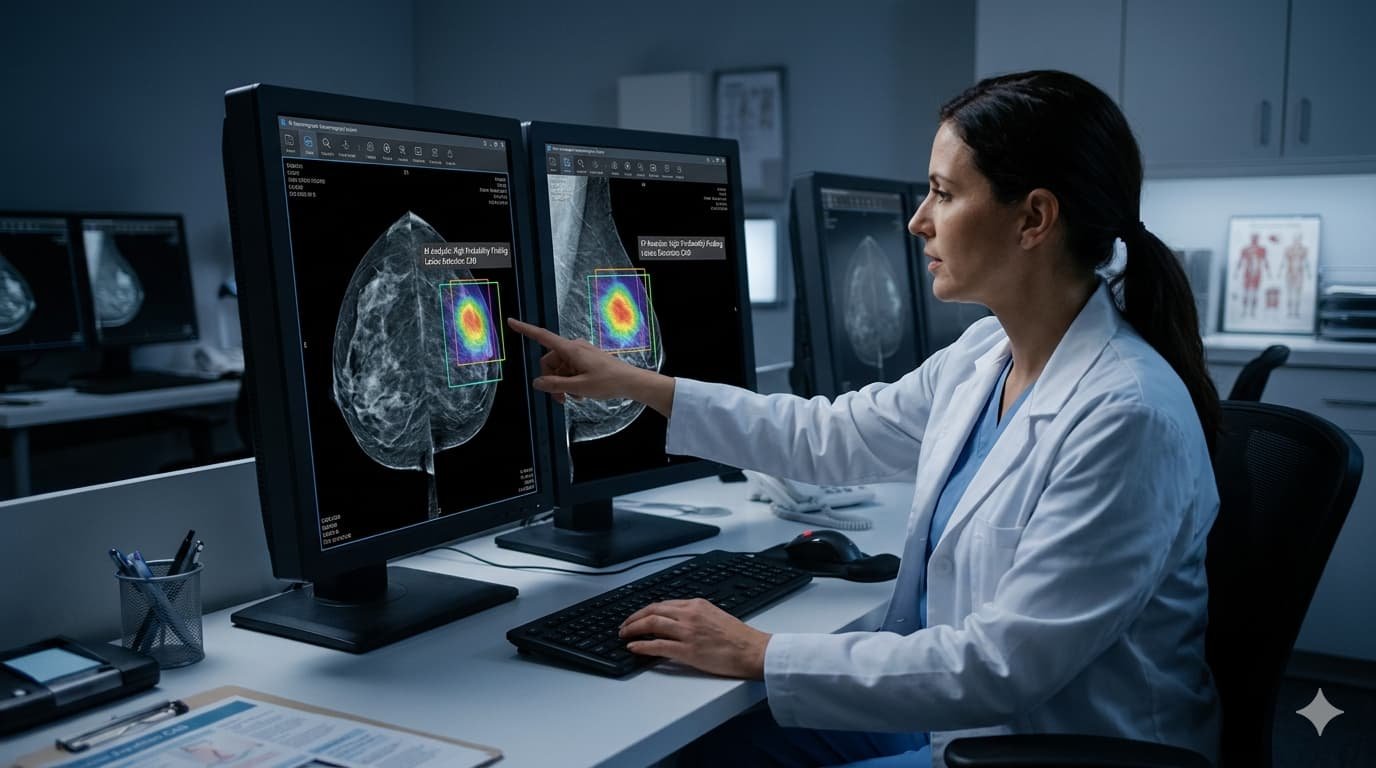
The 175,000-Woman Study That Changed the Conversation
The largest and most rigorous breast cancer AI screening study ever conducted was published this week in the journal Nature Cancer. Researchers from Imperial College London, Google, Cambridge University, and multiple NHS hospital trusts analyzed Google’s mammography AI across two phases — a retrospective study of 115,000 scans from five NHS screening services, followed by a prospective real-world deployment across 12 clinical sites covering an additional 9,000 women.
The results were definitive enough to shift clinical consensus.
Against the performance of a first human reader, the AI achieved higher sensitivity — meaning it caught more cancers — while maintaining non-inferior specificity, meaning it produced no meaningful increase in false positives. The cancer detection rate rose from 7.54 per 1,000 women with human reading to 9.33 per 1,000 with AI — a gain of roughly 24%.
The most striking finding was in interval cancers — tumors that are missed during routine screening and only discovered when symptoms appear between scheduled appointments, typically at a more advanced and harder-to-treat stage. AI identified 25% of interval cancers that human radiologists did not flag at the prior screening. In many of those cases, earlier detection could mean the difference between Stage I treatment and Stage III treatment — a difference measured in survival rates.
For women having their first mammogram — a group that presents unique diagnostic challenges because no prior scan exists for comparison — the AI results were most dramatic: 39.3% fewer unnecessary recall callbacks, and an 8.8% higher cancer detection rate than the human reader. First-time screeners are often younger women who face higher baseline anxiety about the process, making false positives especially psychologically costly.
What It Means for the American Radiology Shortage
The United States is facing a critical and worsening shortage of radiologists — the physicians who read mammograms, CT scans, MRIs, and other medical images to diagnose disease. The shortage has been building for years, driven by a combination of physician retirement, training pipeline constraints, and surging demand as an aging population requires more imaging.
The NHS study found that using AI as a second reader — checking the work of a human radiologist rather than replacing them — reduced the total reading workload by nearly one-third. In a system stretched by staffing shortages, that efficiency gain has immediate practical value: fewer scans waiting for review, faster results for patients, and radiologists freed to spend more time on the complex cases that require the most human judgment.
American radiologists and oncologists who have reviewed the study have generally responded with cautious enthusiasm rather than alarm about displacement. The AI performs best as a collaborative tool — flagging areas of concern that the human reader then examines with additional scrutiny — rather than as a replacement for clinical judgment. In the words of one study author, AI does not replace the radiologist but amplifies the radiologist’s ability to catch what might otherwise be missed.
The FDA has now authorized more than 70 AI-associated medical devices — more than half of them focused on cancer imaging — suggesting the regulatory pathway for clinical deployment is established and accelerating.
MIT’s CleaveNet: A Blood and Urine Test for 30 Types of Cancer
While the mammography study focused on AI improving an existing screening method, MIT researchers published a different kind of advance: an AI system that designs entirely new cancer-detection tools from the ground up.
The system, called CleaveNet, was developed in the lab of MIT’s Sangeeta Bhatia — one of the world’s leading researchers in nanotechnology-based cancer detection. CleaveNet uses AI to design peptide sequences: short chains of amino acids that, when coated onto nanoparticles and introduced into the body, are cleaved — cut apart — by specific enzymes called proteases that cancer cells overproduce.
When those peptides are cleaved, they release signals detectable in urine. The concept transforms cancer detection from a imaging challenge — requiring expensive equipment, trained specialists, and a clinical visit — into a chemistry challenge that could theoretically be performed at home with a test strip.
Previous versions of this nanoparticle sensor technology used a trial-and-error process to find peptides that worked. The problem was that most peptides could be cleaved by multiple different proteases — meaning the signals detected could not be attributed to a specific enzyme, reducing diagnostic precision. CleaveNet solves this by generating peptide candidates that are highly specific to target proteases, enabling sensors that can distinguish between cancer types rather than simply indicating that some elevated protease activity is present.
Bhatia’s lab is part of an ARPA-H funded initiative to develop an at-home diagnostic kit capable of screening for 30 different cancer types in early stages of disease using urine-based protease activity measurements. That kit does not yet exist — the research is at the design and validation stage — but CleaveNet’s ability to generate highly specific peptides on demand is the key enabling technology that makes the broader project feasible.
Why This Week’s Advances Matter Together
Both breakthroughs address the same fundamental problem in cancer medicine: most cancers that kill people were detectable before they killed — but were not detected, either because the right test was not available, the screening infrastructure was not accessible, or the human capacity to read every scan with perfect accuracy does not exist.
The NHS mammography study proves that AI can close the human accuracy gap in existing screening programs — catching cancers that trained physicians miss, and doing so at scale across diverse populations without sacrificing specificity.
CleaveNet points toward a world where cancer screening is not contingent on access to an imaging center, a specialist, or a scheduled appointment. A future at-home urine test for 30 cancer types would democratize early detection in ways that current screening infrastructure — expensive, geographically uneven, and dependent on radiology staffing — cannot.
Neither advance is a cure. Both are tools for catching a disease earlier — which, in oncology, is almost always the most powerful intervention available.