WASHINGTON, MARCH 18, 2026 —
Key Takeaways
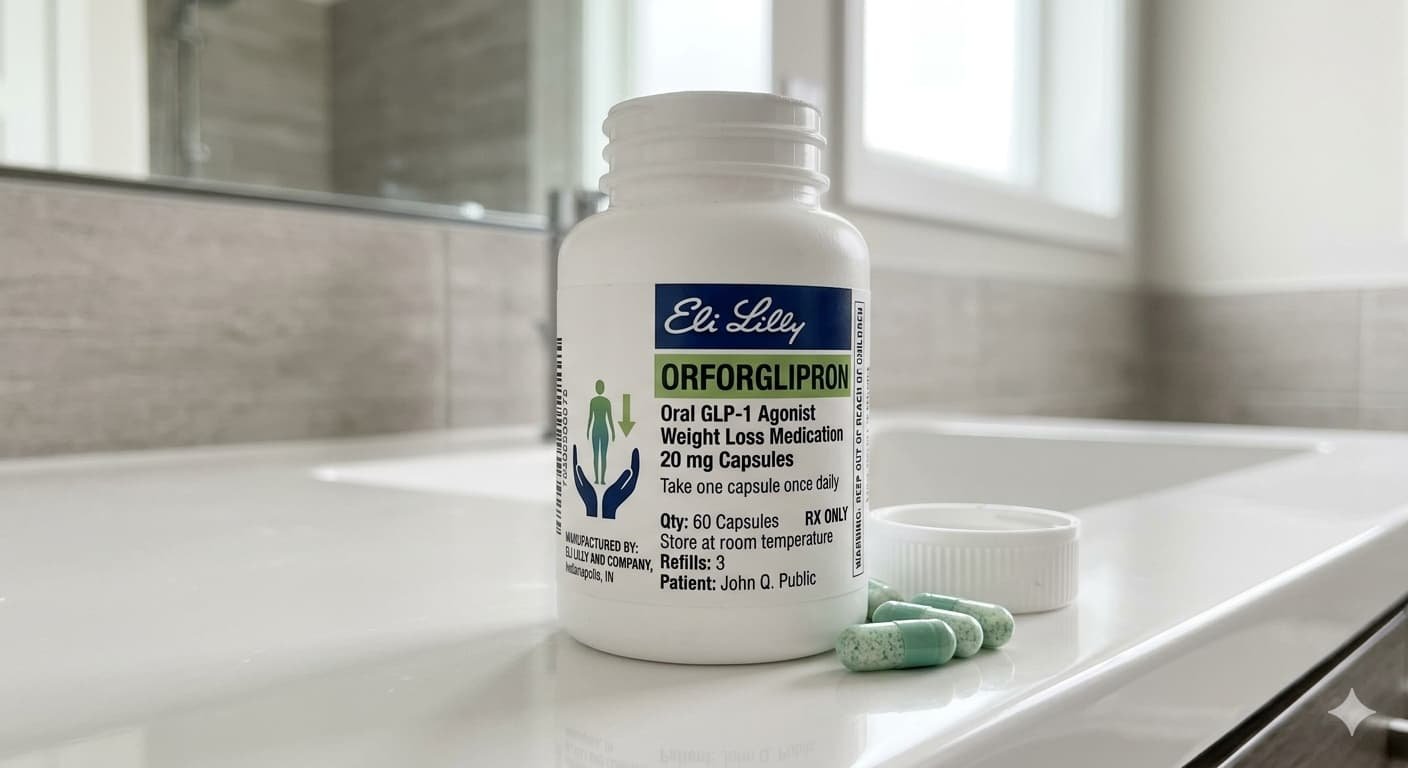
- Eli Lilly’s orforglipron is a once-daily oral weight loss pill awaiting FDA approval in Q2 2026 — no injections, no fasting requirements, no weekly shots
- Phase 3 trials showed patients lost an average of 10.5% to 12% of their body weight — results that rival the convenience of injectable GLP-1 drugs like Wegovy and Zepbound
- Lilly has pledged to price orforglipron at $149 per month for self-pay patients — a fraction of what injectable obesity drugs currently cost
For the past three years, the only people losing significant weight with medication have been the ones willing to inject themselves once a week. Wegovy. Zepbound. Ozempic. The drugs work. But the needle is a barrier for tens of millions of Americans who want the results without the shot.
That barrier is about to come down. Eli Lilly has submitted its application to the Food and Drug Administration for approval of orforglipron — a once-daily oral pill that targets the same biological pathway as injectable GLP-1 drugs — and an FDA decision is expected in Q2 2026. The agency fast-tracked the review under its new Commissioner’s National Priority Voucher program, cutting the standard 10-to-12-month review window to weeks.
What Orforglipron Is — and How It Works
Orforglipron belongs to the same class of drugs — GLP-1 receptor agonists — that made Wegovy and Ozempic household names. GLP-1 drugs work by mimicking a hormone the body produces after eating, signaling fullness to the brain, slowing stomach emptying, and reducing appetite.
What makes orforglipron different is its form. Unlike Wegovy and Zepbound, which are injectable peptide-based drugs requiring refrigerated storage and weekly self-injection, orforglipron is a small molecule pill taken once daily. Critically, it does not require fasting or strict timing before eating — a significant advantage over oral Wegovy, which must be taken on an empty stomach with no more than four ounces of water followed by a 30-minute wait.
In Phase 3 clinical trials, patients taking orforglipron at the highest dose lost an average of 10.5% to 12% of their body weight over 72 weeks. That falls slightly short of Zepbound’s 20% average in trials, but it is a result that would transform the health of most people who achieve it — and the pill format means far more people are likely to actually take it consistently.
How Orforglipron Compares to Other Weight Loss Options
2026 Weight Loss Drug Comparison
| Drug | Type | Avg. Weight Loss | Monthly Cost | Fasting Required |
|---|---|---|---|---|
| Orforglipron | Daily pill | 10.5–12% | $149 | No |
| Oral Wegovy | Daily pill | ~15% | $149 | Yes — 30 min |
| Wegovy (injectable) | Weekly shot | ~15% | $349–$499 | No |
| Zepbound (injectable) | Weekly shot | ~20% | $349+ | No |
The Price That Could Change Everything
One of the most significant developments surrounding orforglipron is not the drug itself — it is the price. Lilly struck a deal with the White House in November 2025, pledging that upon FDA approval, self-pay patients will be able to access orforglipron through Lilly’s direct pharmacy channel with the lowest dose starting at $149 per month.
That price point is dramatically lower than injectable GLP-1 options without insurance, which run from $349 to $499 per month. For Americans who have been priced out of the obesity drug revolution, orforglipron’s pricing could be the moment the market finally reaches them.
Lilly CEO David Ricks has projected the oral drug market could ultimately reach 40 million more Americans who are currently unable to access injectable treatments — either because of cost, needle aversion, or the logistical challenges of cold storage and weekly dosing.
The Maintenance Breakthrough
Beyond initial weight loss, orforglipron has shown a second major use case that has drawn significant attention from doctors and researchers. In the Phase 3 ATTAIN-MAINTAIN trial, patients who had already lost weight on injectable GLP-1 drugs — Wegovy or Zepbound — were switched to orforglipron to maintain their results.
The outcome was significant. Patients who switched from Wegovy to orforglipron maintained nearly all of their weight loss over the following year, gaining less than one kilogram. This matters because one of the biggest problems in obesity treatment is weight regain after stopping injectable drugs — a pattern published research has consistently documented. A daily pill that maintains the results of an expensive weekly shot could be the long-term solution millions of Americans need.
What Stands Between Americans and This Pill
The FDA has not yet approved orforglipron. The agency’s review is ongoing under the National Priority Voucher program, and while an accelerated timeline is expected, no statutory deadline exists. Lilly CEO David Ricks told investors in January that the review was moving at pace and he anticipated a Q2 2026 decision — meaning approval could come as early as April or May.
Once approved, the drug will be available through Lilly’s direct pharmacy platform as well as traditional retail pharmacies and telehealth providers. Medicare coverage for obesity drugs — historically excluded — remains a policy question in Washington, though Lilly has flagged access for Medicare patients as a priority.
The obesity drug market is projected to exceed $100 billion globally by 2030. Orforglipron’s arrival as an affordable, needle-free option could accelerate that timeline significantly — and give tens of millions of Americans their first realistic access to medically supervised weight loss.
What This Means For You
If you or someone you know has avoided injectable weight loss drugs because of cost, needle aversion, or inconvenience — orforglipron is worth watching closely. An FDA decision is expected within weeks to months. Once approved, the starting dose will be available at $149 per month without insurance through Lilly’s direct pharmacy. Talk to your doctor now about whether a GLP-1 medication is appropriate for you, so you are ready to act the moment approval is confirmed.